Human PapillomaVirus (HPV)
| A | Type 16 | |
| B |
Type 18 |
|
| C |
Type 11 |
|
| D |
Option a and b both |
High Risk HPV includes :
| A |
Type 16 |
|
| B |
Type 18 |
|
| C |
Type 11 |
|
| D |
Option a and b both |
Ans. is a and b i.e. Type 16; and Type 18
On the basis of strength of association to Ca cervix – HPV has been classified into
Low risk type
- Cause genital warts and subclinical HPV infection
- Cause high grade CIN which progress to invasive cancer
- Type 16, 18, 31. 33, 45, 58
- Subtype of HPV 6.
HPV associated with Laryngeal Papilloma are?
| A | 13 & 32 | |
| B |
16 & 18 |
|
| C | 6 & 11 | |
| D |
16 & 32 |
6 & 11 REF: Robbins 7th edition page 324
See APPENDIX-31 for “HUMAN PAPILLOMA VIRUS”
Koilocytes with a perinuclear halo on pap smear is pathognomic of?
| A | HPV infection | |
| B |
Metaplasia |
|
| C | Dysplasia | |
| D |
Bacterial vaginosis |
HPV infection
“Koilocytes, dyskeratocytes, macrocytes, etc. are some of the cytologic findings that are characteristic of condylomas (HPV). Koilocytes with perinuclear halo is pathognomonic of HPV infection”
|
Condition |
Type of cell |
|
Estrogen dominated uterus |
Normal cornified cells |
|
Progesterone dominated uterus |
Intermediate cells |
|
pregnancy |
Intermediate cells , Navicular cells |
|
menopausal |
Basal , parabasal |
|
HPV |
Koilocytes, dyskeratocytes, macrocytes with perinuclear halo. |
| A |
HIV |
|
| B |
CMV |
|
| C |
HSV I |
|
| D |
HPV |
Ans. is D. i.e. HPV
Various strains of the human papillomavirus (HPV, notably types 16, 18, 31, 33, 35, and 39) have been implicated as causing a variety of genital lesions, including genital warts, cervical dysplasia, cervical cancer, vaginal dysplasia, vaginal cancer, and penile cancer. The koilocyte is the term used to describe the characteristic of nuclear alterations and perinuclear halo seen with HPV infection in the cervical squamous epithelial cells.
-
Infection with the HIV virus cannot be recognized by cytologic changes.
-
Cytomegalovirus infection causes nuclei to be large, eosinophilic, and “smudged”; cytomegalovirus is uncommonly seen in Pap smears.
-
Herpes simplex I and II infections cannot be distinguished cytologically, as both produce multinucleated cells whose nuclei develop a characteristic “ground-glass” appearance.
| A | Papova virus | |
| B |
Parvovirus |
|
| C |
Herpes virus |
|
| D |
Poxvirus |
Ans. is ‘a’ i.e., Papova virus
-
Papovaviruses are DNA viruses containing double-stranded DNA, are icosahedral in shape, and do not have a lipoprotein envelope. They are commonly found in humans and other species, mostly mammals.
-
Human papilloma virus belongs to the papovaviridae family.
-
Human papillomavirus (HPV) is a viral infection that’s passed between people through skin-to-skin contact. There are over 100 varieties of HPV, more than 40 of which are passed through sexual contact and can affect your genitals, mouth, or throat.
-
Symptoms: Genital wart; Wart; Cervical cancer
-
Diseases or conditions caused: Anal cancer; Head and neck cancer
| A | Monovalent | |
| B |
Bivalent |
|
| C |
Quadrivalent |
|
| D |
Both bivalent and quadrivalent |
Ans. is. ‘d’ i.e., Both bivalent and quadrivalent
Human papillomavirus (HPV) vaccine
- HPV is one of the most important risk factor for cervical cancer, widespread vaccination has the potential to reduce cervical cancer deaths around the world by as much as two thirds, if all women were to take the vaccine and if protection turns out to be long term.
. In addition, the vaccine can reduce the need for medical care, biopsies and invasive procedures associated with the follow up from abnormal pap tests. Thus, helping to reduce the health care costs and anxieties related to abnormal pap tests and follow up procedures.
. HPV vaccines are ‑
1) Preventive vaccines
– The role of HPV vaccine is to prevent infection with certain species of Human papillomavirus associated with the development of cervical cancer, genital warts and some less common type of cancers.
– These vaccines are based on virus like particles (VLPs) assembled from recombinant HPV coat proteins (major capsid protein L1).
– Currently, one quadrivalent product containing HPV types 6,11,16 and 18 has been licenced in US and
recommended by the centres for disease control and prevention for administration to girls and young women
9-26 years of age —> type 16 and 18 are most important as they cause 70% of cervical cancer world wide. – Another product contains HPV types 16 and 18 ( bivalent) and is likely to be available in the near future.
2) Therapeutic vaccines (under trial)
– In addition to above two preventive vaccines, laboratory research and several human clinical trials are focused on the development of therapetic HPV vaccines. In general these vaccines focus on the main HPV oncogenes, E6 and E7. Since expression of E6 and E7 is required for promoting the growth of cervical cancer cells and cells within warts, it is hoped that immune responses against two oncogenes might eradicate established tumors.
| A | Given in women age group 20-40 years | |
| B |
Primary dose consists of 2 doses |
|
| C |
Efficacy > 70% for cervical cancer |
|
| D |
All |
Ans. is ‘c’ i.e., Efficacy >70% for cervical cancer
. There are two types of HPV vaccines :-
i) Quadrivalent :- containing HPV types 6,11,16,18
ii) Bivalent :- containing HPV types 16,18
. Efficacy of vaccine has varied according to the immunologic and virological characteristics of study populations at baseline and according to the endpoints evaluated. Most of the time, rates of vaccine efficacy exceed 90%.
. Vaccine is recommended for girls and young women 9-26 years of age.
. The quadrivalent vaccine is administered intramuscularly as three separate doses, with second dose given at 2months after the first dose and the third dose 6 months after the first dose.
| A | 18,31 | |
| B |
17,12 |
|
| C |
6,11 |
|
| D |
16,18 |
Ans. is ‘c’ i.e., 6,11
. High risk HPV: HPV-16, 18, 31, 33 & 45 —> associated with cervical cancer.
. Low risk HPV: HPV- 6 & 11 –> associated with precursor lesions CM and condyloma accuminatum
. HPV-5 & 8 associated with squamous cell cancer in patients with epidermodysplasia verruciformis
| A | Single papilloma | |
| B |
Multiple papillomatosis |
|
| C |
Osteoma |
|
| D |
Sarcoma |
Ans. is ‘b’ i.e., Multiple papillomatosis
o Multiple warts are common.
Bivalent HPV vaccine contains which types ‑
| A |
Type 6,11 |
|
| B |
Type 6,16 |
|
| C |
Type 16,18 |
|
| D |
Type 11,18 |
Ans. is ‘c’ i.e., Type 16,18
Human papillomavirus (HPV) vaccine
- HPV is one of the most important risk factors for cervical cancer, widespread vaccination has the potential to reduce cervical cancer deaths around the world by as much as two thirds, if all women were to take the vaccine and if protection turns out to be long term.
- Also, the vaccine can reduce the need for medical care, biopsies and invasive procedures associated with the follow up from abnormal pap tests. Thus, helping to reduce the health care costs and anxieties related to abnormal pap tests and follow up procedures.
- HPV vaccines are ‑
1) Preventive vaccines
- The role of the HPV vaccine is to prevent infection with certain species of Human papillomavirus-associated with the development of cervical cancer, genital warts and some less common types of cancers.
- These vaccines are based on virus-like particles (VLPs) assembled from recombinant HPV coat proteins (major capsid protein L1).
- Currently, one quadrivalent product containing HPV types 6,11,16 and 18 has been licensed in the US and recommended by the centers for disease control and prevention for administration to girls and young women 9-26 years of age — type 16 and 18 are most important as they cause 70% of cervical cancer worldwide.
- Another product contains HPV types 16 and 18 ( bivalent) and is likely to be available shortly.
2) Therapeutic vaccines (under trial)
- In addition to the above two preventive vaccines, laboratory research and several human clinical trials are focused on the development of therapeutic HPV vaccines. In general, these vaccines focus on the main HPV oncogenes, E6 and E7. Since the expression of E6 and E7 is required for promoting the growth of cervical cancer cells and cells within warts, it is hoped that immune responses against two oncogenes might eradicate established tumours.
This condition are most commonly caused by which of the following serotypes of HPV
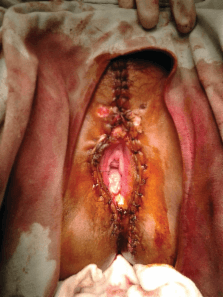
| A |
HPV 6 |
|
| B |
HPV 16 |
|
| C |
HPV 18 |
|
| D |
HPV33 |
This condition is Genital Warts (condyloma accuminata)
Ans A
- Burkitt’s lymphoma (Epstein-Barr virus)
- Hepatocellular carcinoma (hepatitis viruses)
- Cervical cancer [human papillomavirus (HPV)]
- T cell leukemia (retroviruses)
| A |
Condylomalata |
|
| B |
Condyloma acuminata |
|
| C |
Bubo |
|
| D |
Chancre |
Ans. is ‘b’ i.e., Condyloma acuminata
Cervical warts are seen with which HPV ‑
| A |
11,13 |
|
| B |
6, 11 |
|
| C |
17,18 |
|
| D |
5, 8 |
Ans. is ‘b’ i.e., 6, 11
Most common type of HPV associated with cervical cancer ‑
| A |
6, 11 |
|
| B |
5, 8 |
|
| C |
16, 18 |
|
| D |
6, 8 |
Ans. is `c’ i.e., 16, 18
- HPV DNA of oncogenic types (High risk) in HPV-16, 18, 31, 33 and 45 → associated with cervical cancer
- HPV-6 and 11 (Low risk HPV) → associated with precursor lesions of cervical cancer (CIN) and Condyloma Acuminatum.
- In patients with epidermodysplasia verruciformis, Squamous cell cancer develop frequently at sites infected with specific HPV types, including 5 and 8.
- E6 and E7genes of HPV are responsible for carcinogenicity.

