Order Of Kinetics
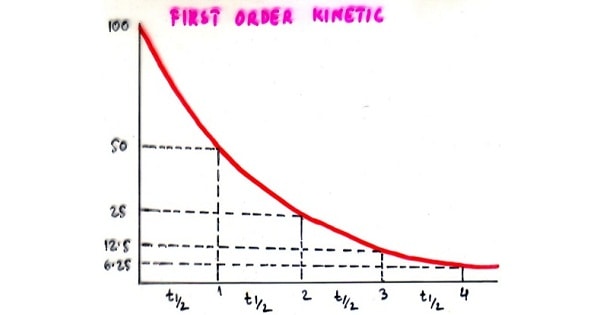
FIRST ORDER KINETICS:
- Absorption, distribution, biotransformation and excretion processes are mostly occurring at rates proportional to plasma drug concentration.
- A constant fraction of drug is absorbed, distributed, biotransformed and excreted per unit time.
- These processes increase in rate with increase in conc. and decrease with falling conc.
- A constant fraction of drug in body is eliminated per unit time.
- Rate of elimination is proportional to amount of drug in body.
- For drugs with first order kinetics time required to achieve steady state levels can be predicted from half life.
- The majority of drugs are eliminated in this way.

| Half life |
Elimination |
| 1 t1/2 | 50% |
| 2 t1/2 |
75% |
| 3 t1/2 |
87.5% |
| 4 t1/2 | 93.75% |
| 5 t1/2 | 96.875 |
FIRST ORDER METABOLISM:
- A drug may be given in doses that produce blood concentrations less than Km of enyzme for drug.
- v = Vmax [C]
- Km + [C]
- When Km >>> [C],
- then v = Vmax [C] & v σ [C]
- Km
- Rate of elimination is directly proportional to drug concentration.
- A constant fraction of remaining drug is metabolized per unit time.
- Most drugs are given at concentrations smaller than Km of enzymes of their metabolism.
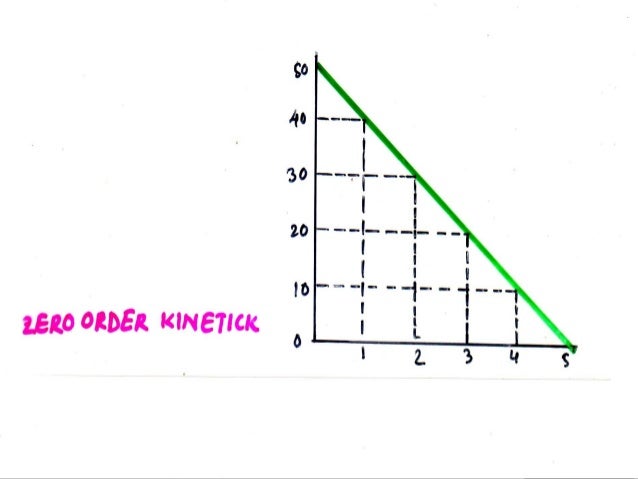
ZERO ORDER KINETICS:
- A constant amount of drug is eliminated per unit time.
- Supply of enzymes are limited in body.
- So, with increasing dose enzymes gets saturated at a particular stage.
- At this point there is,
- No elimination.
- Constant biotransformation.excretion.
- So, rate of processes or reaction is not proportional to conc. or dose.
- Processes also referred as “Rate limited/zero order/saturation kinetics/clinically called “Non-linear kinetics”.
Eg:
- All enzyme mediated processes.
- Drugs involved: Aspirin, ethanol, phenytoin ,propanolol, tolbutamide etc.
ZERO ORDER METABOLISM:
- A drug may be given in doses that produce blood concentrations greater than Km of enyzme for drug.
-
v = Vmax [C]
-
K m + [C]
-
When [C] >>> Km,
-
then v = Vmax [C] , and v = Vmax
-
[C]
A constant amount of remaining drug is metabolized per unit time.
Phenytoin undergoes zero order metabolism at doses given.
Drugs showing zero/pseudo-zero order kinetics :
- Phenytoin
- Theophylline
- Alcohol
- Tolbutamide
- Warfarin
- Salicylates (Aspirin)
- Propanolol
|
First Order Kinetics
|
Zero Order Kinetics (Non linear Kinetics)
|
Exam Question
- Zero order kinetics is independent of plasma concentration.
- Zero order kinetics is otherwise known as saturation kinetics.
- In zero order kinetics constant amount of drug eliminated per unit time.
- Zero order kinetics occur in Phenytoin,Tolbutamide, Propranolol and Ethanol at high dose.
- In first order kinetics a constant proportion of plasma concentration is eliminated.
- In first order kinetics rate of elimination is directly proportional to drug concentration.
- In first order kinetics clearance is constant.
- Elimination after 4 half lives in first order kinetics is 93%.
- Elimination after 3 half lives in first order kinetics is 87.5%.
- For drugs with first order kinetics the time required to achieve steady state levels can be predicted from half life.
- First order kinetics is elimination of the drug is proportional to the serum concentration.
Don’t Forget to Solve all the previous Year Question asked on Order Of Kinetics